Some people with rheumatoid arthritis (RA) take a disease-modifying antirheumatic drug (DMARD) and/or a biologic and reach remission. Others try drug after drug after drug and have yet to find one that’s effective — or that treats their symptoms without causing intolerable side effects. At the same time, other patients with active rheumatoid arthritis opt not to take the most powerful and effective medications on the market or can’t take them because of their medical history or personal risk factors that would make doing so dangerous.
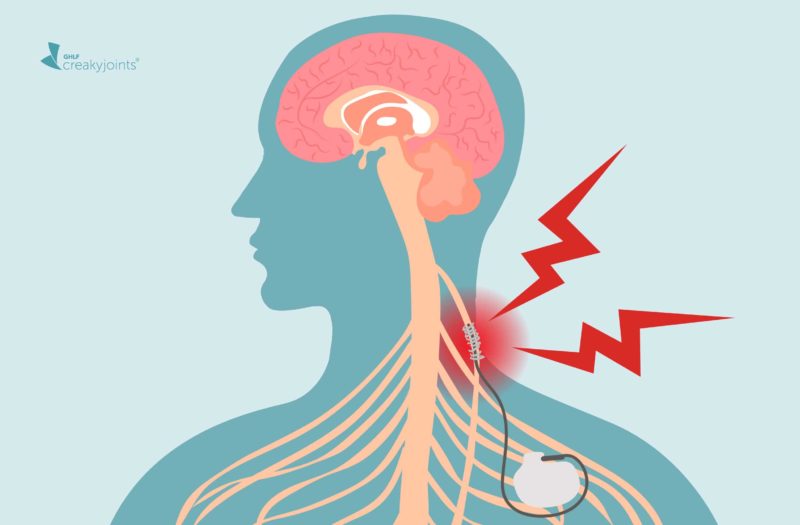
While many scientists are continuing to explore better drug options, some are focused on developing new treatments that aren’t medications at all. One such experimental approach that’s gaining some traction is called neuromodulation, which entails using electrical impulses to stimulate nerves that regulate inflammation in the body.
What Is the Vagus Nerve — and What Does it Have to Do with Rheumatoid Arthritis?
The longest of the cranial nerves, the vagus nerve runs from the brain, through the face and neck, and down into the abdomen. It’s a key part of the autonomic nervous system, the division of your nervous system that controls functions you don’t have to think about, such as breathing, digesting food, and the beating of your heart. The vagus nerve is also the home of the inflammatory reflex, a pathway that appears to be crucial for detecting and modulating inflammation.
When something in your body gets injured or attacked by an invader (like a virus or bacteria), the vagus nerve and the inflammatory reflex help decide how strong of an immune response the body should mount. When that response is appropriate, germs get killed off and injured tissue starts to heal. But when it’s too aggressive for the threat at hand — imagine trying to put out a small candle by holding it under a waterfall — you end up with chronic inflammation, which results in joint and tissue damage.
If you have an autoimmune or inflammatory condition, you already know that your immune system is far more revved up than it should be. In the case of rheumatoid arthritis, the immune system mistakenly tries to attack healthy tissue in the joints as well as in other organs. No one knows exactly what sets RA in motion, but experts do know that inflammatory substances called cytokines play an important role in causing tissue damage. Those cytokines include tumor necrosis factor (TNF) and various interleukins (IL), among others.
Scientists now know that the vagus nerve has the power to reduce the production of these cytokines.
It’s worth noting that the vagus nerve also modulates the body’s stress response and is integral to the mind-body connection, says Vibeke Strand, MD, Adjunct Clinical Professor in the Division of Immunology/Rheumatology at Stanford University and a pharmaceutical and biotech consultant.
This overlap isn’t just an interesting coincidence. “The autonomic nervous system is part and parcel of our immune response,” says Dr. Strand. “It takes a little time for some people to wrap their heads around that.”
While stress alone doesn’t directly cause inflammatory conditions like RA, it can certainly exacerbate them and lead to additional immune system overactivity as modulated by the vagus nerve, Dr. Strand adds.
Emerging research suggests that stimulating the inflammatory reflex — such as by sending electrical impulses to the vagus nerve — has the potential to treat RA by reducing the body’s production of inflammatory cytokines.
Neuromodulation vs. RA Medication: Same Targets, Different Approach
Many people with RA take medications that target specific cytokines. Biologic medications such as adalimumab (Humira), certolizumab pegol (Cimzia), etanercept (Enbrel), golimumab (Simponi, Simponi Aria) and infliximab (Remicade) target the ctytokine TNF, whereas anakinra (Kineret) targets IL-1 and sarilumab (Kevzara) and tocilizumab (Actemra) target IL-6.
But these therapies don’t work for everyone with RA. For these patients, neuromodulation may be another way to reduce the production of cytokines that cause inflammation. If ongoing and future studies are positive, neuromodulation might one day become a viable option for RA patients who don’t respond to medication, can’t take it, or don’t want to, says Dr. Strand. It might also be used in conjunction with drug therapies to boost their effectiveness.
Several researchers and biotech companies are currently investigating neuromodulation for RA. Most are focused on stimulating the vagus nerve.
“The premise is that when you stimulate the vagus nerve it leads to the production of acetylcholine [a neurotransmitter], which binds to receptors on cells that secrete cytokines. When acetylcholine binds to those cytokine-producing cells, it inhibits them from producing TNF and interleukin-6,” says rheumatologist Mark Genovese, MD, Emeritus Professor of Medicine at Stanford University.
Dr. Genovese conducted a pilot study on vagus nerve stimulation that was presented at the 2019 European Congress of Rheumatology (EULAR) conference. The full version of the same research, sponsored by bioelectric company SetPoint Medical, was published in the journal, The Lancet Rheumatology, in 2020.
In the study, 14 people with RA who had tried and did not adequately respond to at least two medications had a small “MicroRegulator” — about the size of a nickel — implanted on the left side of the neck along the vagus nerve. Although the primary goal of this small trial was to assess the safety of the device, half of patients who had impulses sent to the MicroRegulator once a day had significant improvements in their RA disease activity scores. A decrease in cytokine production was also measured.
A few adverse effects were reported, but all were temporary. Those included pain and swelling at the incision site as well as one patient who had temporary vocal cord paralysis.
SetPoint Medical has now begun a larger, multi-center clinical trial designed to learn more about whether vagus nerve stimulation can be used to treat RA, especially in people who don’t respond sufficiently to drugs. This new trial, called RESET-RA, has already begun recruiting participants. The goal is to enroll up to 250 people with RA at multiple sites around the country. To be eligible, patients must have tried and failed several biologic medications or a JAK inhibitor, another kind of targeted therapy for RA.
RESET-RA is a randomized, controlled, blinded study. Everyone who participates will be implanted with a tiny device in the neck during a 45-minute outpatient surgery. The device is about the size of a coffee bean, says David Chernoff, MD, Chief Medical Officer of SetPoint. During the study, only half of the patients will get the device turned on, and participants won’t know whether they’re in the “on” group or the “off” (placebo) group until later. After data is collected, the plan is to turn on the device for everyone in the study, says Dr. Chernoff.
Patients interested in learning more can visit the RESET-RA website.
“If this works, it wouldn’t replace traditional therapies, but it would be an alternative,” says Dr. Chernoff. “Although the drugs we have today are great, they don’t work in all patients and have serious side effects.”
Dr. Chernoff says he is optimistic that the trial will be successful, but that proof can only come in the form of randomized, controlled blinded studies like the RESET-RA trial. He’s also hoping that other companies will follow suit and contribute to the data on neuromodulation — and indeed they are.
Non-Invasive Options for Vagus Nerve Stimulation
While SetPoint is focused on an implantable vagus nerve stimulator, other researchers have been exploring non-invasive approaches to neuromodulation for RA.
In 2019, researchers from the Feinstein Institute for Medical Research, the research arm of Northwell Health, a New York-based health care and hospital system, conducted a small study that found simply holding up a vibrational device to the ear could be used to stimulate the vagus nerve and inhibit the production of cytokines including TNF, IL-1, and IL-6. That research was published in the journal Bioelectronic Medicine.
The following year, in 2020, another small study found that RA patients with high disease activity were able to improve their disease activity scores by applying an external vagus nerve stimulator to the outside of their neck. Participants in that study, which was published in the Scandinavian Journal of Rheumatology, used a device called gammaCore, but the device’s manufacturer has not announced any plans to pursue it as an RA treatment at this time.
More recently, Nesos, another company focused on using electrical signals to treat immune dysfunction, has conducted a small proof-of-concept study using a device that stimulates the vagus nerve externally. In this study, which was published in early 2021 in the journal, The Lancet Rheumatology, 30 patients wore an in-ear device (similar to wireless earbuds) that delivered pulses for up to 30 minutes a day for 12 weeks.
“The vagus nerve has branches, including in the auricle of the ear,” explains Dr. Genovese, who was one of the co-investigators. Each participant got a custom-fit device (researchers took a mold of their ear). “We were able to demonstrate an improvement in common clinical outcomes, including DAS 28-CRP and ACR-20,” he says. (Both are different ways of assessing disease activity in RA.)
Four patients had adverse events during the study period, though only one seemed related to the device itself (minor ear abrasion).
While that sounds promising — especially for a noninvasive treatment — this study was small and did not have a placebo arm. To expand on these early findings, Nesos has launched a larger randomized controlled trial that is currently enrolling participants at multiple sites across the U.S., says Nesos founder Konstantinos Alataris, PhD.
This new study, called PROTECTION, is open to RA patients who have had an incomplete response to the DMARD methotrexate but have not yet tried a biologic or JAK inhibitor. Participants will receive electrical pulses through earbuds worn for 15 minutes each day; the control group will wear the earbuds but not receive the pulses.
RA patients who are potentially eligible and interested in participating in PROTECTION can ask their rheumatologist to refer them to a participating site, says Dr. Alataris.
Another Approach to Neuromodulation: Splenic Nerve Stimulation
While most players in the field of neuromodulation for RA are experimenting with different ways to stimulate the vagus nerve, Galvani Bioelectronics — a joint venture between GlaxoSmithKline (GSK) and Verily (Google’s life sciences company) — is instead targeting the splenic nerve.
The splenic nerve is another part of the inflammatory reflex. As with SetPoint’s neuromodulation device, Galvani’s potential treatment would involve outpatient surgery. In this case, however, the stimulating device would be implanted in the spleen, which resides in your upper abdomen.
“The vagus nerve connects with the splenic nerve, which obviously goes to the spleen; that’s where the nervous system and immune system meet,” says Kristoffer Famm, PhD, President of Galvani. Dr. Famm believes that getting as close to this juncture as possible would lead to higher efficacy. He adds that stimulating the splenic nerve is also likely to be safer than vagus nerve stimulation, which may carry a risk of heart-related side effects, such as chest pain.
Galvani is currently enrolling qualified RA patients in a research study testing their implanted device. To be eligible, patients must:
- Be 22 to 75 years old
- Have active RA
- Have adult-onset RA for at least six months
- Have had an inadequate response to at least 2 biologic DMARDs and/or JAK inhibitors, including at least one TNF inhibitor
- Have an appropriate washout from previously used biological DMARDs or JAK inhibitors
- Be receiving treatment with standard dose(s) of conventional synthetic DMARD(s)
According to the company, all study-related medical care will be provided at no cost; compensation will also be provided for time and travel. To see if you qualify, click here.
Looking Ahead: The Future of Neuromodulation
While this all might sound a bit futuristic, neuromodulators are already on the market — just not yet for rheumatoid arthritis.
Implantable stimulators have been FDA-approved for patients with drug treatment-resistant epilepsy and depression since 1997. The same gammaCore device that Scandinavian researchers tested for RA has FDA clearance in the U.S. to treat migraines and cluster headaches, and it currently has FDA Emergency Use Authorization to treat COVID-19-related breathing problems.
Meanwhile, vagus nerve stimulation is being investigated for a wide range of potential applications including inflammatory bowel disease, lupus, bipolar disorder, and Alzheimer’s disease.
“Just like there was a wave during the past two to three decades of targeted biologics and synthetic therapies, there could be a wave of neuromodulation devices,” says Dr. Famm.
Be a More Proactive Patient with ArthritisPower.
Join CreakyJoints’ patient-centered research registry to track your symptoms, disease activity, and medications — and share with your doctor. Read more and sign up here.
Addorisio ME, et al. Investigational treatment of rheumatoid arthritis with a vibrotactile device applied to the external ear. Bioelectronic Medicine. April 17, 2019. doi: https://doi.org/10.1186/s42234-019-0020-4.
Aranow C, et al. Transcutaneous auricular vagus nerve stimulation reduces pain and fatigue in patients with systemic lupus erythematosus: a randomised, double-blind, sham-controlled pilot trial. Annals of the Rheumatic Diseases. April 12, 2021. doi: https://doi.org.10.1136/annrheumdis-2020-217872corr1.
Beyer M. Vagus nerve stimulation may reduce the symptoms of rheumatoid arthritis. Medical News Today. June 4, 2019. https://www.medicalnewstoday.com/articles/325532.
Bonaz B. Is-there a place for vagus nerve stimulation in inflammatory bowel diseases? Bioelectronic Medicine. April 3, 2018. doi: https://doi.org/10.1186/s42234-018-0004-9.
Breit S, et al. Vagus Nerve as Modulator of the Brain–Gut Axis in Psychiatric and Inflammatory Disorders. Frontiers in Psychiatry. March 13, 2018. doi: https://doi.org/10.3389/fpsyt.2018.00044.
Drewes AM, et al. Short-term transcutaneous non-invasive vagus nerve stimulation may reduce disease activity and pro-inflammatory cytokines in rheumatoid arthritis: results of a pilot study. Scandinavian Journal of Rheumatology. October 13, 2020. doi: https://doi.org/10.1080/03009742.2020.1764617.
Genovese MC, et al. Safety and efficacy of neurostimulation with a miniaturised vagus nerve stimulation device in patients with multidrug-refractory rheumatoid arthritis: a two-stage multicentre, randomised pilot study. The Lancet Rheumatology. July 28, 2020. doi: https://doi.org/10.1016/S2665-9913(20)30172-7.
Interview with David Chernoff, MD, Chief Medical Officer of SetPoint.
Interview with Konstantinos Alataris, PhD, Founder of Nesos.
Interview with Kristoffer Famm, PhD, President of Galvani.
Interview with Mark Genovese, MD, Emeritus Professor of Medicine at Stanford University and former consultant for SetPoint and Nesos.
Interview with Vibeke Strand, MD, Adjunct Clinical Professor in the Division of Immunology/Rheumatology at Stanford University and a pharmaceutical and biotech consultant.
Kopp WJ, et al. Autonomic nervous system reactivity to positive and negative mood induction: The role of acute psychological responses and frontal electrocortical activity. Biological Psychology. March 2011. doi: https://doi.org/10.1016/j.biopsycho.2010.12.003.
Marsal S, et al. Non-invasive vagus nerve stimulation for rheumatoid arthritis: a proof-of-concept study. The Lancet Rheumatology. February 1, 2021. doi: https://doi.org/10.1016/S2665-9913(20)30425-2.
Nichols JB, et al. Pseudoanginal chest pain associated with vagal nerve stimulation: a case report. BMC Neurology. April 20, 2020. doi: https://doi.org/10.1186/s12883-020-01693-5.
Pavlov V, et al. The vagus nerve and the inflammatory reflex—linking immunity and metabolism. Nature Reviews Endocrinology. November 21, 2012. doi: http://doi.org/10.1038/nrendo.2012.189.
Vagus nerve stimulation. Mayo Clinic. https://www.mayoclinic.org/tests-procedures/vagus-nerve-stimulation/about/pac-20384565.