Learn more about our FREE COVID-19 Patient Support Program for chronic illness patients and their loved ones.
In May, the U.S. Centers for Disease Control and Prevention (CDC) announced that children between the ages of 12 and 15 were eligible to receive the Pfizer COVID-19 vaccine. The news came as a relief to parents and guardians of this age group, hoping the extra protection would allow their children to enjoy some of the socialization and freedoms they’d missed over the past year.
But for parents of younger children who were not yet eligible, the news brought about anxiety and questions. Mainly, when will children younger than 12 be able to get a COVID-19 vaccine? As summer passed and schools planned for in-person learning, parents, guardians, teachers, and others became more eager to have the vaccine available for all children.
Now, it seems the U.S. is getting closer to making that happen. Pfizer-BioNTech has submitted data to the U.S. Food and Drug Administration (FDA) in hopes of receiving emergency use authorization (EUA) for its vaccine to be given to children ages 5 to 11, according to NBC News. Dr. Anthony Fauci, the nation’s leading infectious disease expert, said he is hopeful that this age group will start receiving vaccinations by the end of October.
Still, progress is being made to get more Americans vaccinated, which is especially crucial for people who are immunocompromised and may have a lower response to the COVID-19 vaccine than others. The more people who can get vaccinated against COVID-19, the more protected high-risk groups will be.
So, it’s no surprise that immunocompromised people, regardless of whether or not they are parents to young children, are happy and relieved that a COVID-19 vaccine may soon become available to more people.
The Global Healthy Living Foundation’s COVID-19 Patient Support Program aimed to gain more insight into how its immunocompromised and high-risk patient community feel about a COVID-19 vaccine becoming available for children ages 5 to 11 — particularly those who have or are with children in this age range on a regular basis.
Of the 749 survey respondents (95 percent report being fully vaccinated), 10 percent are a parent or guardian of a child between the ages of 5 and 11, while 29 percent spend time regularly (at least a few hours a week) with children in this age group.
Here are some key learnings and insights from the poll.
The vast majority of immunocompromised respondents who are parents or guardians plan on getting their child vaccinated against COVID-19.
When people who are parents or guardians were asked if they planned on getting their child/children the COVID-19 vaccine once it became available to their age group, 80 percent of respondents said they would.
A free response section where respondents could elaborate on their answers provided insight into why 20 percent of people said they do not plan on getting their child/children vaccinated. (Keep in mind, this was a small number of respondents: only 14.)
Common themes included concerns that the vaccine was being released too soon and questions about long-term side effects.
“It will be critical for pediatricians and public health and infectious disease experts to address these concerns — supported by data from the vaccine trials and shared as part of the authorization process — to help ensure parents and guardians feel safe and comfortable getting their children vaccinated,” says Daniel Hernandez, MD, Director of Medical Affairs and Hispanic Outreach for GHLF.
Even patients who plan to get their child vaccinated have some concerns but think the protection against coronavirus outweigh any potential side effects. As one wrote, “I’m worried that my kids will have bad side effects like I did, but it’ll be well worth it.”
Having the COVID-19 vaccine available to more people makes immunocompromised people feel more protected.
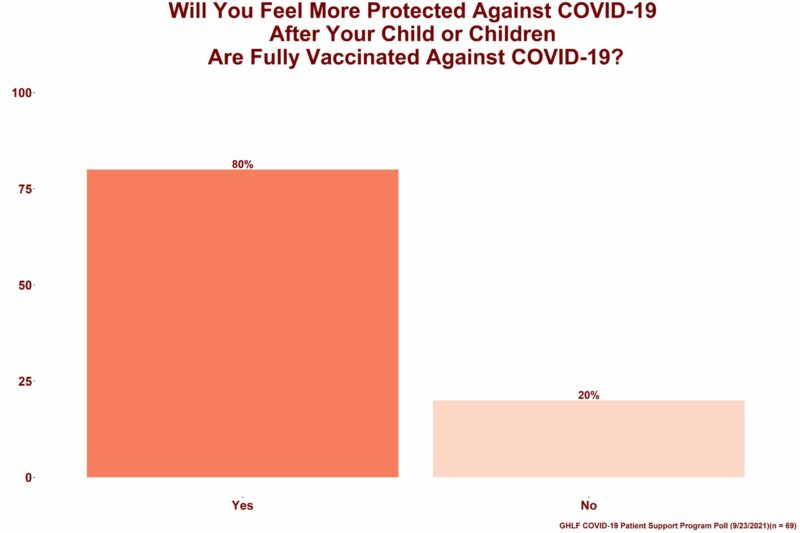
As part of the survey, respondents who are parents or guardians were asked if they would feel more protected against COVID-19 once their 5 to 11-year-old children were vaccinated. Of those, 80 percent said yes.
It is important to remember that the COVID-19 vaccine does not make a person fully immune to the virus. Even fully vaccinated people can become infected with coronavirus (called a breakthrough COVID-19 infection) — and initial research shows that people who are immunocompromised are more likely to experience breakthrough infections than those who are not. This is why giving the vaccine to more people — in this case, children — is an important step to protecting high-risk individuals.
Many respondents expressed these sentiments in a free-response section of the survey.
“I’m immunosuppressed and vulnerable to severe COVID-19,” one person wrote. “Having my child finally vaccinated may lessen the chance of me — and her — getting very sick.”
“I am immunocompromised and my child is as well due to her having JIA [juvenile idiopathic arthritis],” another person shared. “This is a scary time for our family, and we are hoping that the vaccine for children will help alleviate some of our fears while protecting our family.”
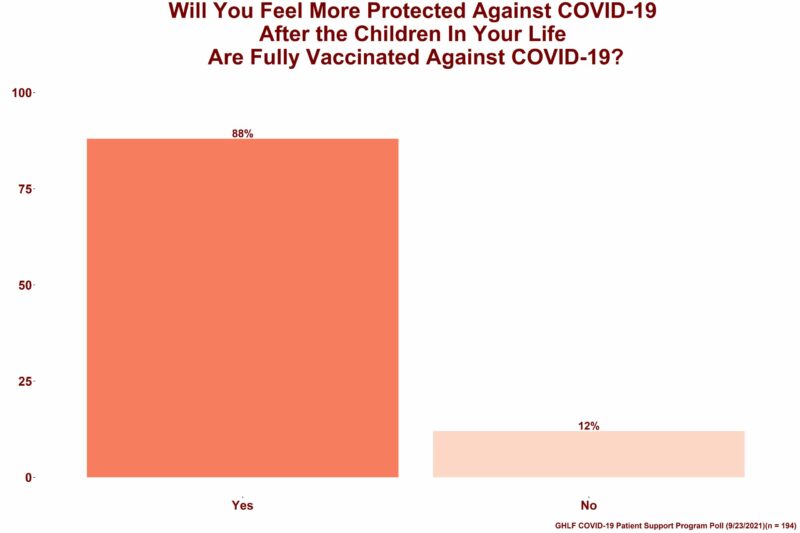
The same question was asked to people who are not parents or guardians but regularly spend time around children (such as a relative, person living in multi-generational household, caregiver, friend, teacher, bus driver, etc.) Similarly, 88 percent said they will feel more protected once the COVID-19 vaccine becomes available to 5- to 11-year-olds.
For grandparents and other relatives, the vaccine gives them a chance to be with family again. “I want very badly to be able to be more involved in my grandchildren’s lives and if they are vaccinated, I will feel much safer around them as an immunocompromised person,” one respondent shared.
Several educators shared that they would feel safer at work once this age group becomes vaccine eligible. “Being a teacher who is immunocompromised I fear everyday working with kids who can’t wear their mask properly,” one respondent wrote. “This will help.”
Another school employee shared similar thoughts, writing “the vaccine becoming available [to more kids] is a huge relief for all of us.”
Many immunocompromised people — whether they are around children or not — are hopeful, happy, and relieved that a COVID-19 vaccine will soon be available to children.
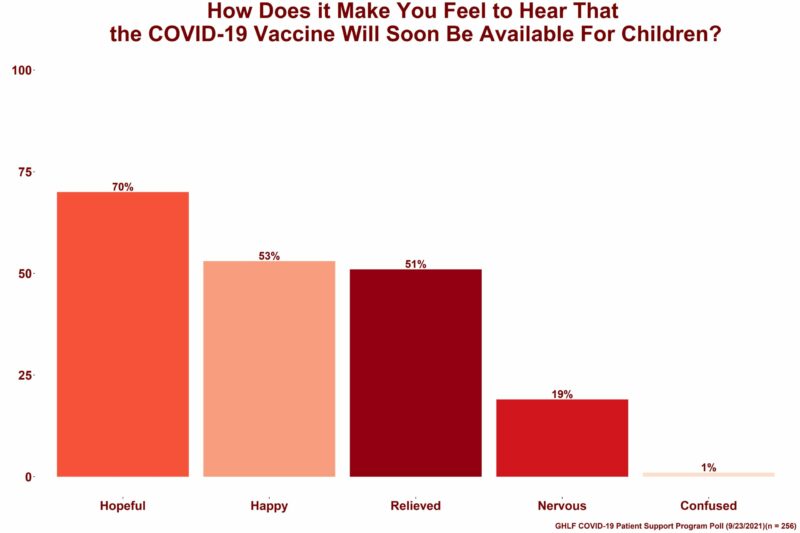
As part of the survey, all respondents were asked to select all the emotions they felt knowing the COVID-19 vaccine would soon be available to children between the ages of 5 and 11. Of those who responded:
- 70% felt hopeful
- 53% felt happy
- 51% felt relieved
- 19% felt nervous
- 1% felt confused
- 1% felt none of the previously listed emotions
For many, the comfort comes from knowing the children in their life could soon have some protection against COVID-19.
“I have been so worried for my 9-year-old grandson,” one person shared. “I can’t be more pleased that now all these children can be protected.”
“We are in Florida and precautionary measures are minimal in our district, so any additional layer of prevention is very welcome, and we are so looking forward to it,” another respondent wrote.
Even those who are not around children regularly acknowledge what this news means for the larger community.
“Don’t discount this as being just for kids,” one person wrote.
“I think vaccinating children is a crucial step in making the threat of this virus manageable,” one patient shared. “It will protect everyone to a higher degree if kids are vaccinated and will help limit the emergence of variants.”
Some immunocompromised people are concerned that parents won’t get their children vaccinated.
But among the happiness and hope, concern lingers. Several respondents are worried about vaccine side effects and safety; many others are worried about not enough children getting vaccinated once the vaccine becomes available to them.
“I am doubtful many parents will get their kids vaccinated,” one respondent shared.
Another had similar thoughts: “I don’t think parents will rush to get kids vaccinated because it is only emergency use authorization.”
One patient shared their concern about mitigation efforts decreasing as more people become eligible for the vaccine.
“I am relieved that this age group will hopefully be able to be protected against serious disease sooner than expected, but I am still concerned that this announcement will result in even lower mitigation enforcement in schools and businesses, and at the state and federal levels,” they wrote. “It’s causing me to keep my kids out of things that they want to do, which feels so unfair to them when they see lots of other kids doing all of these things.”
Of course much remains to be seen about how the rollout of the vaccine for younger children will impact the trajectory of the pandemic, but at least it’s another critically important step in stopping the spread of COVID-19 and resuming more aspects of pre-pandemic life.
“The children have been so patient through all of this,” one respondent wrote. “I am thrilled that they are soon to be protected.”
Currently no COVID-19 vaccines have been approved for children under the age of 12 in the U.S. When the vaccine becomes available, be sure to talk to your child’s health care provider about any questions or concerns you may have.
The Global Healthy Living Foundation is committed to providing ongoing education about COVID-19 vaccines for the chronic illness and immunocompromised community.
To stay informed about the latest COVID-19 vaccine news for people who are immunocompromised, take immunosuppressant medications, or have autoimmune conditions, follow all of our COVID-19 vaccine coverage here.
Get Free Coronavirus Support for Chronic Illness Patients
Join the Global Healthy Living Foundation’s free COVID-19 Support Program for chronic illness patients and their families. We will be providing updated information, community support, and other resources tailored specifically to your health and safety.
About the Patient Support Program Quick Poll
Members of our program have underlying health issues — such as inflammatory arthritis and other autoimmune conditions, heart disease, lung disease, diabetes, and more — that may increase their risk for COVID-19 complications. They are interested in understanding the best ways to stay safe during the pandemic and to be part of a community of people with similar concerns, questions, and fears.
We regularly poll members, who live in the U.S. as well as around the globe, about a variety of topics, including how the pandemic is affecting their lifestyle, mental health, chronic disease management, medication adherence, and more.
We use this information to inform the educational resources we provide and to inform other stakeholders — such as public health experts, policymakers, advocacy groups, health care professionals, and pharmaceutical companies — about chronic illness patients’ needs and concerns. You can participate in ongoing polls by joining the support program here.
ACIP Presentation Slides: July 22, 2021 Meeting. Advisory Committee on Immunization Practices. U.S. Centers for Disease Control and Prevention. July 22, 2021. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-07/07-COVID-Oliver-508.pdf
COVID-19 Vaccines for Children and Teens. COVID-19. U.S. Centers for Disease Control and Prevention. August 17, 2021. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/recommendations/adolescents.html.
Li, DK. Pfizer submits data to FDA, seeking to use its Covid vaccine for children ages 5 to 11. NBC News. September 28, 2021. https://www.nbcnews.com/news/us-news/pfizer-seeks-fda-authorization-covid-vaccine-children-ages-5-11-n1279939.