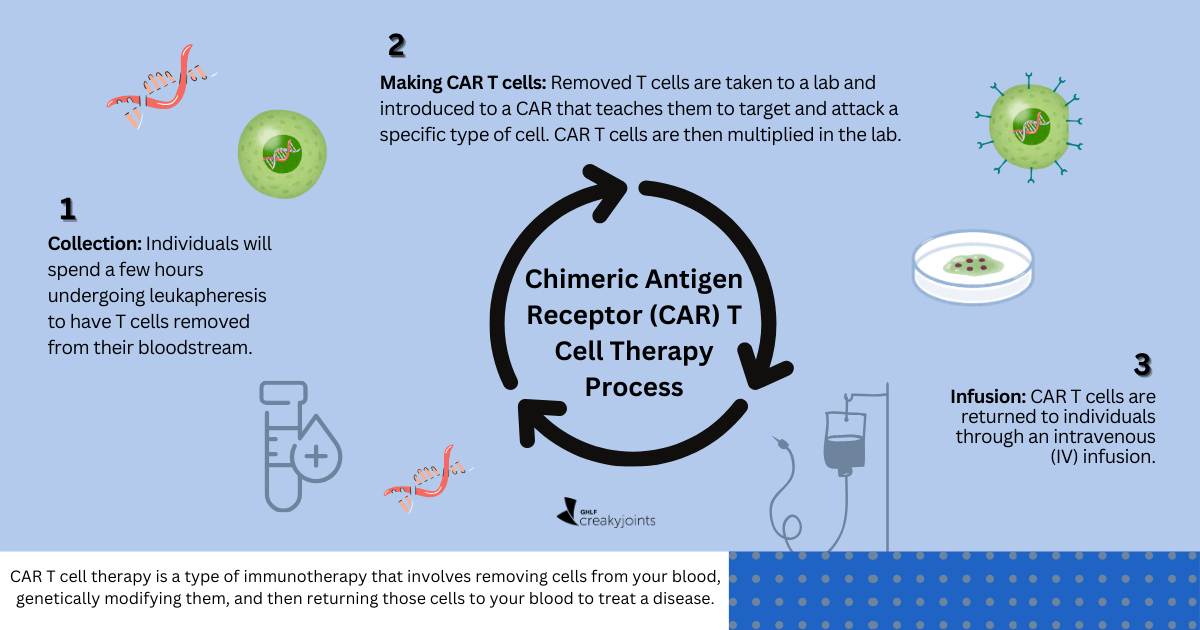
Clinical trials are a crucial step forward in medical advancement. Think of them as the behind-the-scenes work that helps make sure new medicines and techniques — from medication and devices to procedural changes or lifestyle alterations — are safe and effective before they’re available to everyone.
Yet despite the amazing possibilities of clinical trials, several misconceptions exist that can hinder participation. For one, clinical trials are often complex, leading to misunderstanding on how they are conducted, their purpose, and the safeguards in place to protect participants. There is also a historical distrust due to past abuses in medical research along with sensationalized media coverage. Lack of awareness also plays a role, with a gap of communication between the scientific community and the public.
Here, we bust some of the most common myths about clinical trails so you can understand them better and ultimately learn how they can help to improve patient care and enhance the overall patient experience.
Myth: Clinical Trials Are Only for Drugs, Not Devices
A common misconception is that clinical trials are only for testing medications. In reality, clinical trials may test a medical, surgical, or behavioral treatment. There are a variety of purposes for clinical trials beyond testing a medication or a new use for a medication, including:
- Biologics like vaccines and gene therapies
- Surgical procedures
- Behavioral interventions like counseling
- Preventive measures like dietary supplements
- Diagnostic tests such as C-reactive protein
- Nutritional interventions including specific diets
- Rehabilitation programs like physical therapy
- Medical devices (anything that treats a patient without medication)
Myth: Clinical Trials Involve Medications Never Before Used in Humans
Not exactly, says nurse and IgG4-related disease patient advocate Donna Shipp. “A lot of people think that every clinical trial involves something (usually a medication) that has never been used in humans before,” she explains.
While this is true for some medications, such as chemotherapeutic drugs, it is not true for all. Donna emphasizes that a lot of trials are pursued to find out if an existing medication can also be used to treat other conditions. “The critical basic steps of just determining the safety of taking the drug have already been done in the original trial,” says Donna, “So, some of the worry about risk can be mitigated right there.”
Some clinical trials explore the potential use of medicines originally developed for one disease to treat other diseases. This approach is known as drug repurposing or repositioning, where researchers investigate whether existing drugs, already approved for one medical condition, may have beneficial effects for different diseases. By leveraging existing drugs, this strategy can expedite the development process and reduce costs compared to creating entirely new medications, as the safety profiles of these drugs are often already established. Drug repurposing has been successful in identifying new treatment options for various conditions, providing a more efficient way to address medical needs.
Myth: I’ll Be Like a Guinea Pig
“The guinea pigs already passed the trial, and that’s why we moved on to people,” says Donna, adding people get paid more, “but don’t let the guinea pigs know that.”
Humor aside, safety is the top priority in phase one of a clinical trial. Each phase of the clinical trial process serves a distinct purpose, with safety measures primarily assessed in phases one and two. By participating in a phase three clinical trial (the pivotal phase for assessing medical devices) you wouldn’t be among the first group of humans testing the medication, treatment, or device.
Clinical trial participants receive regular monitoring and medical care throughout the study. Researchers closely track participants’ health and address any concerns promptly. Ethical guidelines serve the dual purpose of protecting the well-being of patients and volunteers while maintaining the integrity of scientific research.
Informed consent is a crucial process where participants are informed about key aspects of a clinical trial, and this ongoing communication ensures that individuals can make informed decisions throughout the study.
While safety is paramount, participating in clinical trials carries certain risks, such as:
- Experiencing adverse effects from the treatment, which could range from discomfort to severe or even life-threatening consequences.
- The study may demand more time and energy compared to standard treatments. This includes additional visits to the study site, more frequent blood tests, additional procedures, extended hospital stays, or adherence to complex dosage schedules.
More information about the ethical guidelines and risks is available through NIH Clinical Research Trials and You.
Myth: Clinical Trials Are Only for Very Sick People Who Have No Options Left
Clinical trials cover a wide range of conditions, from common diseases to rare disorders, and they often seek participants at various stages of health. Researchers study different health problems, enrolling both people with specific health issues and those who are generally healthy. Some trials focus on treating existing health problems, while others require volunteers without such issues. Regardless of health status, all participants receive carefully planned treatment and researchers compare their results to see how well the treatments work.
Myth: Once You Start a Clinical Trial, You Can’t Leave
Joining a clinical trial is your choice, and you can decide to leave at any time without facing any consequences or affecting your regular health care from your doctor. If doctors or researchers believe it’s no longer best for you to continue in the trial, they may also remove you, but they will always explain the reasons for doing so. Your well-being is a top priority throughout the process. A participant might decide to quit the clinical trial if the differences between treatments may be greater than they anticipated or if they experience adverse side effects.
Myth: Patients Must Be Invited to Apply for a Clinical Trial
Don’t wait for an invitation. Although doctors may not know of applicable clinical trial options available to all patients, it does not hurt to ask. There are websites that house the open or in-progress clinical trial options. You can search for studies currently recruiting, or are soon to be recruiting, in your area at clinicaltrials.gov or through a national registry like this list from the National Institutes of Health. If you see a clinical trial you want to apply for, talk with your doctor about it about how it might impact your current treatment plan.
Myth: Participation in a Clinical Trial Means You’ll Get a Placebo
A placebo is something that looks like the real treatment but doesn’t actually do anything in your body. It could be a pill without any medicine. Trials use placebos when there’s no standard treatment available. Placebos aren’t used if there’s already an effective treatment, or if it would put you at risk.
Before joining a trial, they will let you know if placebos are used as part of the informed consent process. Your safety and understanding are top priorities throughout the study. Placebos are not used with cancer patients as this is deemed unethical. “Placebos would be used when there is no good active treatment for the patient,” explained Geoffrey Shapiro, MD, PhD, Director of the Early Drug Development Center at Dana-Farber, in a recent video for Dana-Farber Cancer Institute.
When you volunteer for a clinical trial, you might not always receive the treatment being tested. They use a method called a randomized controlled trial, where volunteers are randomly assigned to either get the new treatment (the test group) or a placebo or the current standard care (the control group). Researchers then compare the effects in each group to see if the new treatment works.
You might be in the test group or the control group when you join a trial. Those in the control group don’t get the experimental treatment, but they are crucial for comparison. This helps scientists determine if the new treatment is better than the standard or no treatment. Sometimes, you won’t know which group you’re in until the trial is over, as this helps prevent bias in the results.
Myth: Once Denied, Always Denied
If you are denied participation in a specific clinical trial, you can explore the possibility of applying for other clinical trials. Each clinical trial has specific eligibility criteria, and being denied entry into one trial does not preclude someone from participating in another, provided they meet the criteria for the new trial.
Different trials may have different inclusion and exclusion criteria based on the nature of the research and the goals of the study. Several factors can influence eligibility, including:
- Medical history, including comorbidities
- Current health status
- Age
- Other specific requirements outlined by the researchers conducting the trial
Be a More Proactive Patient with PatientSpot
PatientSpot (formerly ArthritisPower) is a patient-led, patient-centered research registry for people living with chronic conditions. You can participate in voluntary research studies about your health conditions and use the app to track your symptoms, disease activity, and medications — and share with your doctor. Learn more and sign up here.
The Basics. National Institutes of Health. https://www.nih.gov/health-information/nih-clinical-research-trials-you/basics.
Cancer Mythbusters: Cancer Clinical Trials. Dana-Farber Cancer Institute. https://www.dana-farber.org/health-library/cancer-mythbusters-cancer-clinical-trials.
National Library of Medicine. https://www.clinicaltrials.gov/.